CellVantage™ AAV

AAV production enhancer powered by VSE™ technology. Compatible with multiple AAV serotypes, suspension HEK293 cell lines, and PEI transfection reagents, it delivers 2–4× higher titers to increase usable virus output without adding complexity. Read More
Best for: PEI-based transfection and teams seeking a straightforward yield boost.
Simply drop into existing workflows. No Tweaks. Just Titers.
Have any questions? Contact an Expert
| Specifications | |
|---|---|
| Cell Line | HEK293-based |
| Application | AAV Production |
| Transfection Reagents | Optimal for PEI-based |
| Culture Type | Suspension |
| Appearance | White Powder |
| Stability | 24 months |
| Storage | 20 ± 5ºC, protect from light |
| Production System | Transient |
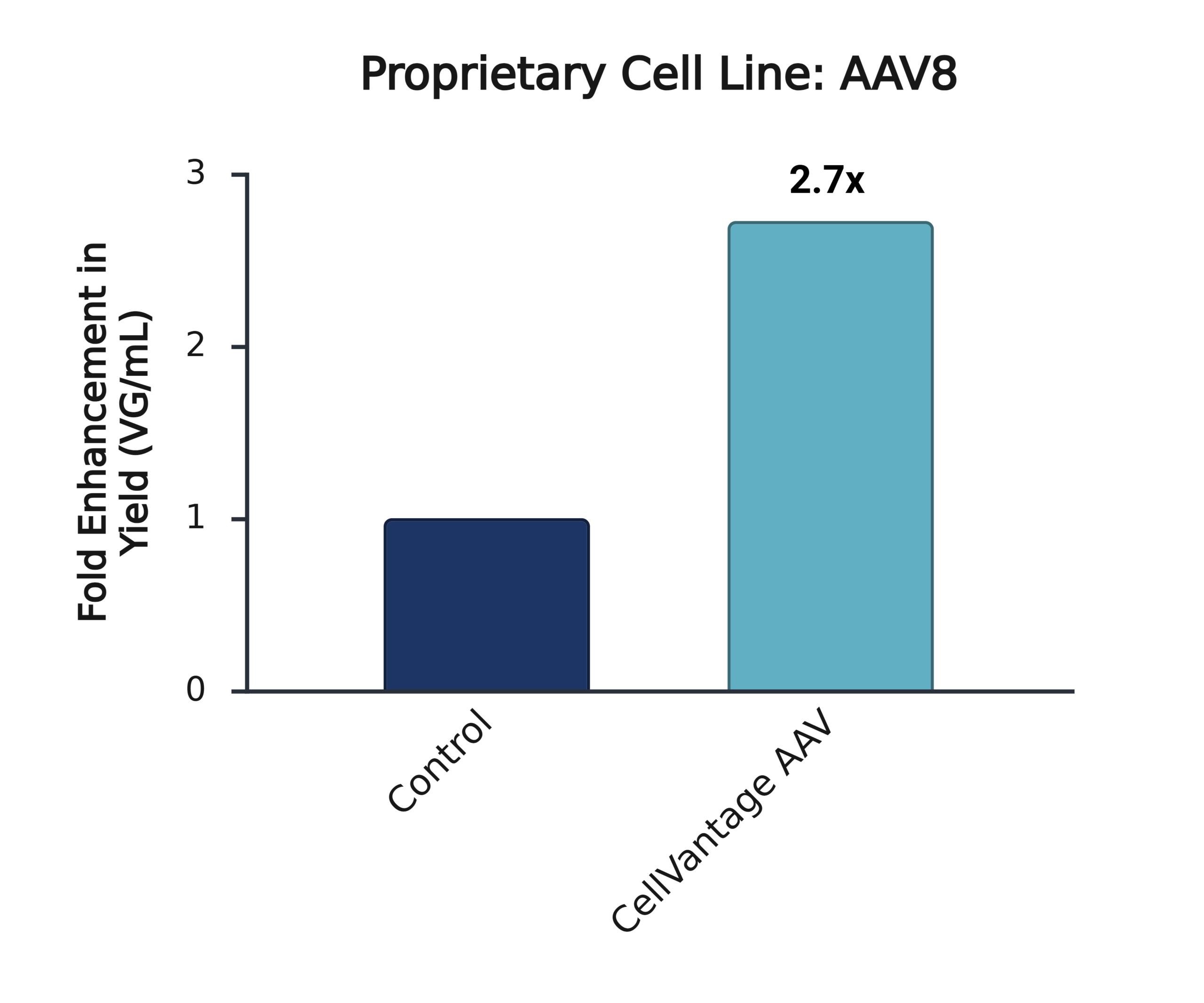
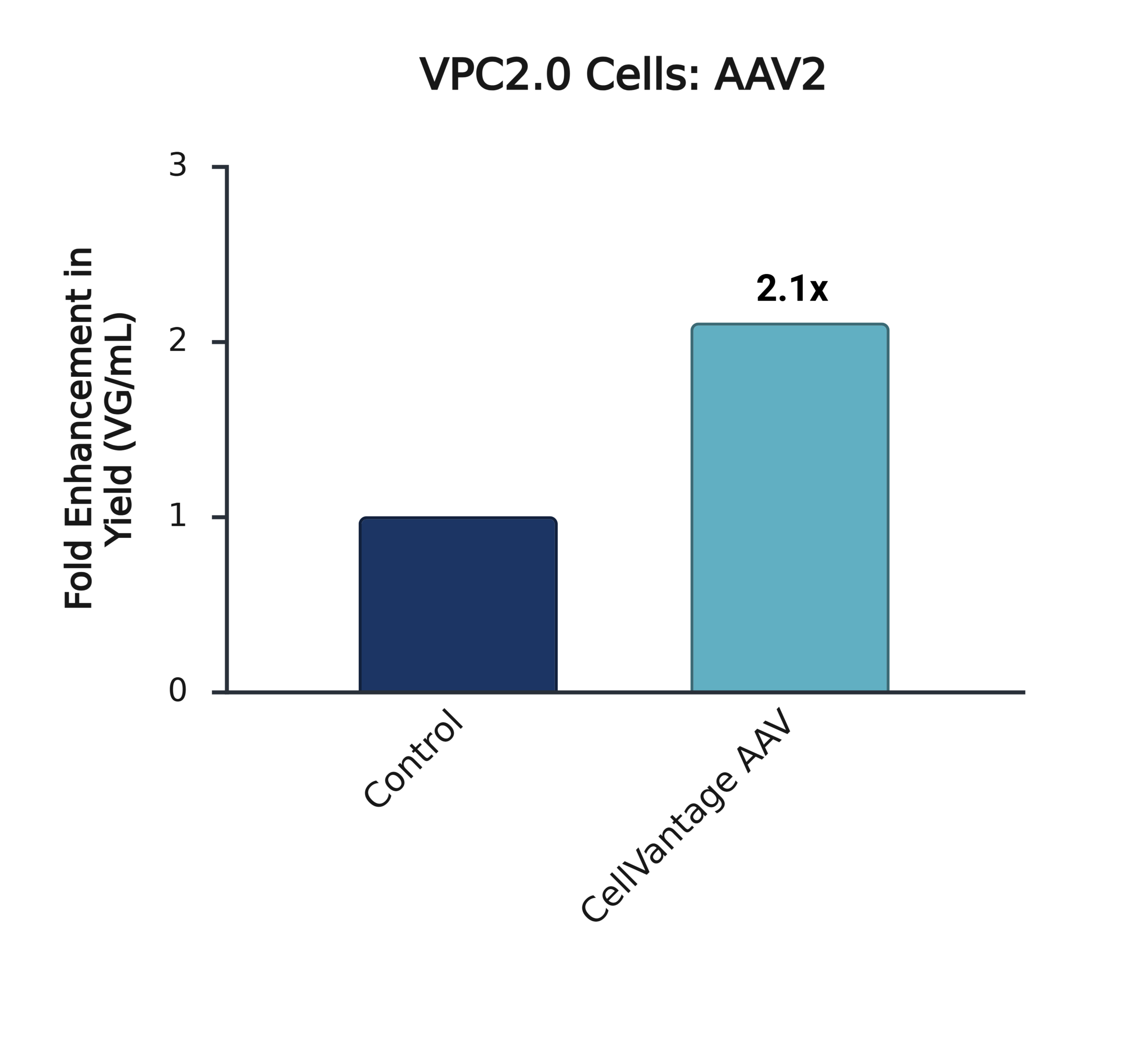
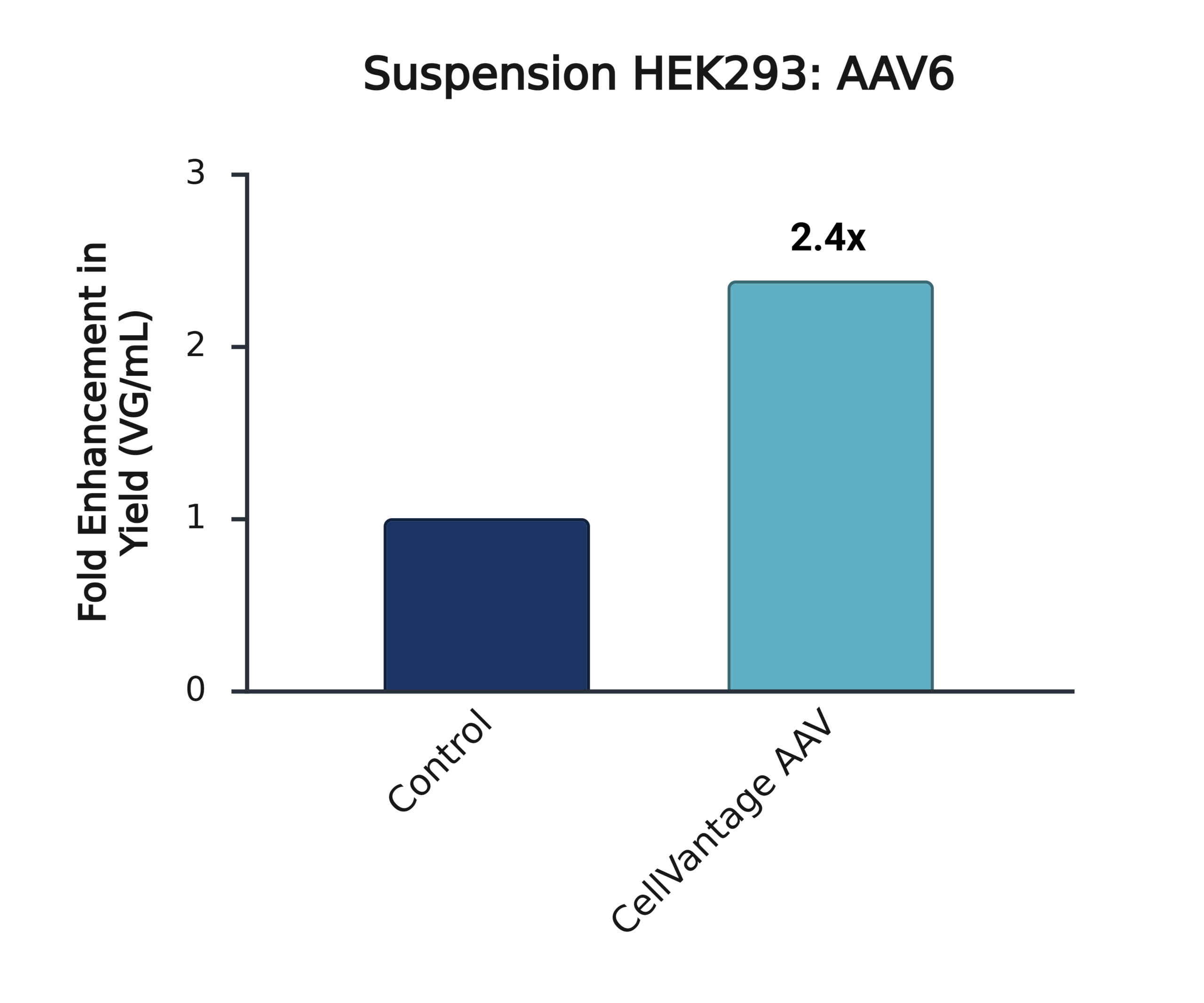
Addition of CellVantage AAV consistently increases AAV production by >2x across multiple HEK293 lineages and AAV serotypes.
A single vial of each enhancer can treat up to 3L of production media at the recommended starting dose. Production volume may vary depending on the optimal dose for a given manufacturing system.
There is no specific solvent provided with CellVantage™ AAV. For dissolving purposes, any 100% tissue culture grade DMSO can be used. For specific inquiries, please contact Virica’s technical team directly.
If required, CellVantageTM AAV may be diluted immediately prior to use in a solution between 100% and 50% DMSO (v/v in media). This can be done to increase pipettable volume and/or minimize DMSO carryover into cell culture. These intermediate working stocks should be be prepared immediately before use and promptly added into the final application. Freezing or reusing working stock solutions is not recommended.
CellVantage™ AAV was specifically designed to improve AAV manufacturing in suspension HEK293-based systems. For other viral vector applications, please contact Virica to discuss the best-fit enhancer from our portfolio. Contact an Expert
Yes. Please contact Virica directly for information on bulk order formats for larger manufacturing scales, as well as GMP availability and pricing.
For typical transfection-based processes, CellVantage™ AAV is administered once and remains in the media throughout upstream production. Re-addition should only be considered if a full media exchange is performed within 4 hours of transfection complex addition. Please consult Virica for application-specific guidance or Contact an Expert.
No, CellVantageTM AAV can remain in the media for the duration of viral production
Research-grade CellVantage™ AAV is not routinely tested for sterility. It should be dissolved in a sterile environment, such as a biological safety cabinet (BSC). Sterile filtration is not recommended or required when testing dose ranges during small-scale formulation development. If sterile filtration is required, please consult Virica for recommended filter types before proceeding.
CellVantageTM AAV should be added at the time of transfection for transient workflows. Where needed, addition within a ±30-minutes of transfection is generally acceptable.